Hypoperfusion
Chronic brain hypoperfusion is a known cause of AD. In humans, MMD is a model of early stage AD with all the hallmarks of this disease. Hypoperfusion of the brain independently induces cognitive impairment and AD. It can also result in End tidal CO2 deficiency. However, the exact mechanisms underlying this condition are unclear. Nevertheless, MMD can be a useful model of chronic hypoperfusion of the brain.
Hypocapnia
In an orthostatic intubation study, significant hypocapnia was observed. This is due to hyperventilation and can be reversed by CO2 rebreathing. However, it is not known whether this effect occurs in all patients with orthostatic intolerance. Therefore, the study’s findings need to be validated in human populations. To this end, Novak will attempt to distinguish between two distinct groups of patients who present with unexplained exercise intolerance.
In acute respiratory failure, increased pCO 2 results in a lowered cerebral perfusion. This condition may arise from several causes, such as pulmonary edema, asthma, and chronic obstructive pulmonary disease. Acute respiratory acidosis may also be caused by a drug overdose or an allergic reaction. In animals, experiments with mild hypercapnia have shown that no adaptive mechanisms suppress the increase in cerebral blood flow (CBF). However, in a clinical hypercapnic population, the pCO 2 value rises to 30 mm Hg or more.
Acute brain injury patients may experience both types of symptoms. While the most common cause is acute brain injury, pulmonary disease may also be the cause of the problem. In such cases, it may be necessary to reverse the disease as soon as possible. Symptomatic treatment with steroids and anti-anxiety drugs may be helpful. But for this, a patient should be admitted for observation and evaluation. A proper diagnosis is crucial.
End tidal CO2
The effect of hypoperfusion due to end tidally CO2 has been studied in acute trauma patients. It has shown to be a prognostic factor compared to other vital signs in certain conditions. In a retrospective study of a single EMS agency, 103 patients with hypotension and shock were studied. They were measured on arrival and at 30minute intervals for up to 120 minutes. They were also measured for respiratory rate and oxygen saturation. Also they were treated according to standard protocols and their survival was monitored for 30 days.
The most reliable way to evaluate the condition of the patient is by monitoring the end tidal CO2 levels. This is the most effective method of determining whether the ETT has been placed correctly. Expiratory CO2 can provide valuable information about metabolic activity, physiologic factors, and ventilator function. It has also been used to predict the outcome of resuscitation.
Carotid artery occlusion
In this case, hypoperfusion due to carotid occlusion occurred on the left side of the patient. Using a vagus nerve model, the carotid arteries were bluntly separated and fastened with 5-0 silk sutures. The patient developed restlessness and increased blood pressure eight hours after the procedure, and left limb hemiplegia was diagnosed 24 hours later.
After right internal carotid artery stenting, a 68-year-old male developed restlessness, left limb hemiplegia, and headaches. A computerized tomography perfusion was performed 24 h after the procedure and revealed abnormal hypoperfusion on the right hemisphere. Transcranial Doppler was performed to evaluate flow velocity in the right middle cerebral artery and anterior cerebral artery. These findings suggested an intracerebral steal phenomenon. Hypervolemic therapy was instituted and the patient’s symptoms resolved within 3 days.
CMP usually occurs in the internal carotid artery, although it can occur in other carotid arteries, including the middle cerebral artery. Although there are some other causes of CMP, these are the most common. Patients with carotid artery occlusion have a significantly increased risk of developing WSI. Although the incidence of CMP increases with angioplasty, most studies have not yet identified a definitive cure.
Migraine
Some studies have suggested that cerebral hypoperfusion can contribute to migraine headache. These findings are based on studies that have shown regional cerebral blood flow in migraineurs during prodrome and headache attacks. A randomized double-blind study of patients with similar characteristics is needed to determine if hypoperfusion is a cause of migraine. Hypoperfusion and migraine may coexist for some period of time, but the relationship between the two is not clear.
A study by Henry PY in the 1950s showed that cluster headache and migraine patients with vascular disease also had cerebral hypoperfusion. The researchers also found that the condition was associated with the presence of aura. Previously, the researchers believed that carotid artery puncture was a trigger for migraine. However, they later found that the procedure had no effect on migraine headaches. However, they did find that patients with this condition were more prone to developing migraine than people without the disease.
The researchers have found that migraine patients who have aura have an increased risk of cerebellar infarction. In addition, prolonged cortical hypoperfusion during an aura may increase the risk of cerebellar infarction. The abnormal perfusion may result in cross-cortical diaschisis, which causes damage to cerebellar tissue. However, these findings need to be interpreted in the context of current knowledge about migraine pathogenesis.
Chronic hypoperfusion in AD
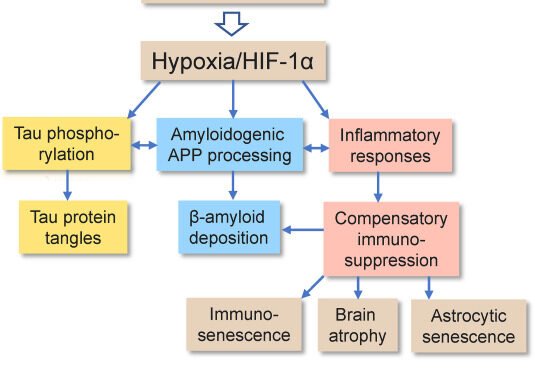
In Alzheimer’s disease, there is a persistent deficit in cerebral blood flow that contributes to the progression of the disease. The brain receives decreased blood supply at rest and has altered perfusion of activated areas. Several mechanisms may contribute to this deficit, including altered neurovascular coupling and diseased tissue. Regardless of the underlying cause, chronic hypoperfusion may worsen the disease and make treatment more difficult. This article will discuss the potential treatment options for patients with AD.
Transgenic mouse models have been useful in the study of neurovascular dysfunction in AD. In transgenic mice, over-expression of the mutated human APP protein causes cerebrovascular dysfunction, and the mice develop cognitive and behavioral symptoms associated with AD. In contrast, mice expressing WT proteins failed to develop Ab deposits. These findings may suggest therapeutic targets for vaccine trials in human AD. However, further research is needed to better understand the mechanism of chronic hypoperfusion.
Mods
MODS is characterized by the failure of normal gas exchange, as indicated by arterial hypoxemia. There are multiple pathologic factors contributing to this impaired gas exchange, including atelectasis, increased capillary permeability, increased diffusion distance of oxygen, and regional injury. Ventilatory support is not a panacea, however, because it can aggravate the lung injury and further atelecta in dependent lung zones.
The underlying cause of MODS is ineffective tissue perfusion, which leads to tissue death. When the circulation does not meet the tissues’ oxygen needs, the organs begin to fail. The patient is often not aware of this problem. However, the loss of consciousness is a symptom of MODS. It is important to note that it is possible to reverse some of the damage caused by MODS, but treatment options are limited.
Symptoms
Hypoperfusion refers to an under-supply of blood to an organ. In severe cases, hypoperfusion can lead to physical problems. It can lead to pain and cell death. Although the underlying cause is unknown, hypoperfusion is a serious health issue. The signs and symptoms of hypoperfusion are described below. The symptoms may include a sudden drop in blood pressure, cool extremities, cyanosis, and altered mental status.
Cerebral hypoperfusion occurs when the blood supply is decreased in specific parts of the brain. This decrease in blood supply can cause dizziness, headaches, nausea, and fainting. In severe cases, blood flow to certain parts of the brain is impaired, leading to cognitive impairment and attention disorders. This condition also has implications for a person’s emotional state. As a result, it is vital to diagnose hypoperfusion early to prevent further damage to the body.
Patients with a high SBP present with symptoms of hypoperfusion. The majority of these patients are warm-wet and discharged from the emergency department. However, the number of patients with hypoperfusion increases significantly in geriatric, psychiatric, and intensive care wards. This is likely related to their haemodynamic profiles on arrival at the ED. It should be noted that not all patients with a high SBP are in cardiogenic shock.
Mechanisms
There are four major categories of mechanisms of hypoperfusion, although there can be more than one in the same patient. Each mechanism is characterized by changes at the cellular level. Here we’ll look at how hypoperfusion occurs in the heart and what the symptoms of each type are. Hypovolemic shock is the result of reduced circulating blood volume, leading to tissue hypoperfusion and cellular dysfunction. To correct this, compensatory mechanisms are activated to maintain intravascular pressure and blood flow to critical organs.
Chronic cerebral hypoperfusion activates a compensatory vascular mechanism that tries to restore CBF to optimal levels. Unfortunately, this mechanism fails to prevent neuronal death and cognitive impairment. Because neurons begin dying before CBF is restored, pharmacological intervention is necessary to boost cerebral blood flow. For example, a compound isolated from the seeds of Chinese celery, dl-3-nbutylphthalide, is shown to enhance CBF and prevent neuronal loss. Further, dl-3-nbutylphthalide has been shown to reduce inflammation and ameliorate memory deficits in acute ischemic animal models.